Interdisciplinary Initiatives Program Round 7 - 2014
Mary Teruel, Chemical & Systems Biology
Sanjay Lall, Electrical Engineering, Aeronautics & Astronautics
Allison Okamura, Mechanical Engineering
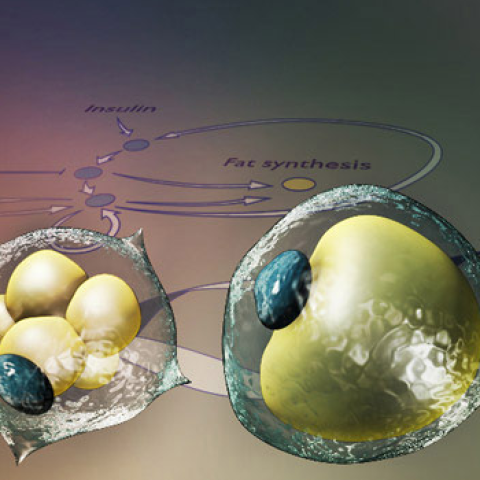
The overall goal of this proposal is to understand how glucocorticoid oscillations link to fat cell production. Glucocorticoids are a powerful tool to treat many human illnesses, including autoimmune diseases and cancer, and to prevent rejection following organ transplantation. Inhaled glucocorticoids are considered the most effective longterm treatment for asthma, a disease which in 2011 afflicted 235–300 million people globally and resulted in 250,000 deaths. Glucocorticoids have been shown to be highly effective for treating rheumatoid arthritis, one of the most common and serious forms of arthritis, causing chronic pain and a high risk of disability and mortality. While use of glucocorticoids has many beneficial effects, their use also leads to obesity, insulin resistance, diabetes, and osteoporosis. These harmful side effects are closely linked to production of too many or dysfunctional fat cells resulting from the fact that glucocorticoids potently stimulate mesenchymal stem cells to differentiate into fat instead of bone.
We have recently uncovered key molecular mechanisms that control fat cell production and developed the first quantitative molecular model of the fat cell differentiation process (1). In particular, we showed for the first time that the production of fat cells from precursor cells is controlled by a bistable switch that is driven by multiple feedback loops between critical regulator proteins and PPARG, the master transcriptional regulator of fat cell differentiation. Intriguingly we have evidence that only certain frequencies of stimuli can be transmitted through the bistable switch to result in fat cell production. This powerful filtering feature of the bistable switch is what we in the Teruel Lab now want to explore with the help of two experts in control theory – Dr. Sanjay Lall, an Associate Professor in the Depts. of Electrical Engineering and Aeronautics and Astronautics, and Dr. Allison Okamura, an Associate Professor in the Depts. of Mechanical Engineering and Computer Science. If we can understand what types of glucocorticoid stimuli can pass through the bistable switch and result in fat cell production versus which are filtered out, we would have an important means to deliver glucocorticoids to human patients for therapeutic purposes while preventing the harmful side effects resulting from glucocorticoid-driven fat cell production.