
Photo courtesy of the Kurtzig family: Jamie Kurtzig, 12, holds some of her hybrid closed-loop system equipment.
Stanford Medicine News Center - April 5th, 2017 - by Andrew Schwartz
At 19 months old, Jamie Kurtzig was diagnosed with Type 1 diabetes. For the next 10 years, her parents would wake up every three hours during the night to prick their daughter’s finger so they could check her blood glucose level. If her blood glucose was too low, they gave her food to avoid seizures or a loss of consciousness. If it was too high, they gave her an insulin injection to bring the level down to a normal range.
“It’s caused a kind of PTSD for my husband and me,” said Sara Kurtzig, who lives with her daughter and husband in Marin, California.
But for the past year, they’ve been able to sleep through most nights. That’s because Jamie started using a hybrid closed-loop insulin delivery system in 2016, thanks to a clinical trial at Lucile Packard Children’s Hospital Stanford and Stanford Medicine that assessed the system’s use in children ages 7 to 14.
“The closed-loop system has completely changed our lives,” Sara said. “It took me a month to trust it, but now I can go to bed at 11 p.m. and wake up at 6:30 a.m. almost every night.”
The system is among the methods being tested by researchers at the School of Medicine and Lucile Packard Children’s Hospital in their efforts to find easier ways for younger children with Type 1 diabetes to get the doses of insulin they need.
Bruce Buckingham, MD, professor of pediatric endocrinology, directs clinical trials of the closed-loop system, which modulates insulin delivery based on glucose sensor readings measured every five minutes. He called the system a “historic advance” for diabetes care.
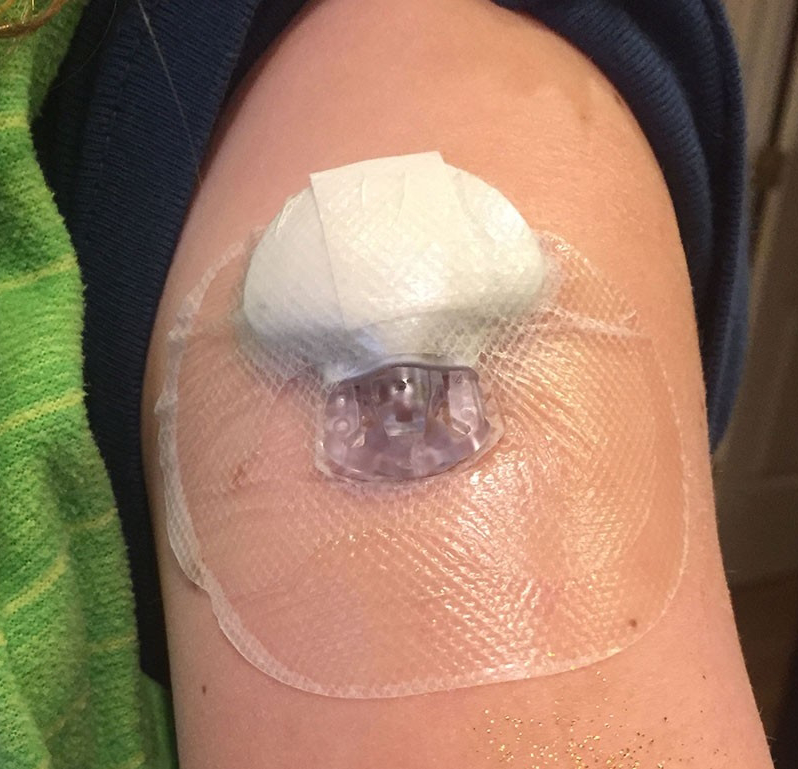
Photo courtesy of the Kurtzig family: The sensor inserted into Jamie’s
arm monitors her glucose levels and communicates the data to her
insulin pump. She is especially happy it can stay on while swimming.
“With this system, patients can achieve very reliable and safe overnight glucose control, mitigating overnight highs and lows with minimal manual intervention,” said Buckingham, who treats patients at Packard Children’s. The improved glucose control dramatically decreases the risk for overnight seizures and long-term complications associated with Type 1 diabetes.
Type 1 diabetes is an autoimmune disease in which the body’s immune system attacks insulin-producing cells in the pancreas. As a result, the pancreas produces little or no insulin, a hormone that brings glucose from the bloodstream into the body’s cells to be used as energy. Without insulin, the body cannot use glucose as energy. Too much insulin can cause severe low blood glucose levels, which can result in seizures, loss of consciousness and, in worst-case scenarios, death. Too little insulin can lead to high blood glucose levels and long-term complications. That’s why people with Type 1 diabetes have to frequently check their glucose levels.
Clinical trials lead to FDA-approved devices
In September 2016, an article in the Journal of the American Medical Association detailed the successful multicenter trial of a hybrid closed-loop insulin delivery system for patients with Type 1 diabetes over the age of 14. Later that month, the FDA announced approval of the device tested in the study, the Medtronic MiniMed® 670G system, for that age group.
The system, commonly referred to as an artificial pancreas, works by wirelessly linking an insulin pump and a glucose monitor. While some of the testing and blood-sugar adjustments can be made by the system, patients must still perform these tasks themselves prior to eating.
Buckingham, a co-author of the article, receives research support from Medtronic. He noted that Stanford conducted the initial studies on this system at a camp for children with diabetes in 2014.
“We are not yet to the point where these systems have been tested in all age groups or where they truly mimic all functions of a human pancreas, so there is more work to do,” said Buckingham. Among the challenges: Current hybrid closed-loop systems still require patients to assess the amount of food (carbohydrates) they are eating and to deliver an insulin dose through their pump before meals.
Buckingham and his closed-loop team at Stanford continue to work toward improving the system. Their efforts include testing and adapting these devices for younger children as well as testing systems with different user interfaces and different methodologies that adjust for exercise and insulin delivery at meals.
Helping younger patients and their families
The hybrid closed-loop system has other advantages, as well. Twelve-year-old Jamie Kurtzig, now old enough to want the freedom to do things like attend sleepovers at a friend’s house, said, “When the system is in auto mode, it monitors my blood sugar every five minutes and keeps up the proper basal rate [of insulin]. Now I only have to check blood sugar four times a day. In the past, I had to check it a lot more, even up to 12 times a day.”
Buckingham noted that the advance wouldn’t be possible without the willingness of people like the Kurtzigs to participate in trials. “We have been very fortunate to have a diabetes community that’s interested in doing studies and collaborating with us,” he said.
Knowing that the device is not yet FDA-approved for children younger than 14, Sara Kurtzig is committed to the importance of participating in trials.
“We’ve had such a positive experience and have reached a new level of stability in Jamie’s glucose control. So if we can play a part in making this technology available to all Type 1 diabetes patients, it feels like we are really making a difference,” she said.
Jamie blogs about her experience and said the device has given her both freedom and responsibility she didn’t have before. “I do more things for myself now, but I still have to deliver insulin manually when I’m eating carbs because it doesn’t do that on its own yet,” she said.
The Kurtzigs believe the improved control Jamie has seen with her closed-loop system has been worth the pricks, pokes and inconveniences that come with it. “I wanted to sleep better and I wanted my mom to sleep better. I also really wanted to help other families and to contribute to science all around the world. Because of the trial, I really feel like I get to be a part of diabetes history,” Jamie said.
Refining the systems
The goal of hybrid closed-loop systems is to make patients’ diabetes care less of a burden and to keep their glucose values in a safe range so they can be healthier. To make further progress toward this goal, the Stanford team has been part of a multicenter, NIH-funded study group that is trying to eliminate the need for patients to give themselves an insulin dose (or bolus, as it is known in the diabetes world) before eating — an onerous task, particularly when it has to be done at every snack and every meal.
“In today’s closed-loop systems, the insulin comes on a little slower and lasts a little longer than we would like,” said Buckingham. “Those lag times make it difficult to provide insulin delivery for a meal in a full closed-loop system. We are looking forward to working with fast-acting insulins — and more rapid delivery — to improve meal glucose control and decrease the daytime burden of diabetes.”
To that end, Stanford is the only institution involved in four National Institute of Diabetes and Digestive and Kidney Diseases research projects, which begin in the 2017-18 fiscal year. The projects will test multiple automated, closed-loop devices in what could be the final steps before requesting regulatory approval for permanent use.
Korey Hood, PhD, professor of pediatrics and of psychiatry and behavioral sciences at the School of Medicine, will lead the pediatric diabetes psychology research team that is investigating how to best help children and their families use these systems, and is partnering with Buckingham on the research.
“Part of our mission is to ensure that the system will be used properly by young patients, meaning that it has the desired impact on both a patient’s health and quality of life,” Hood said. “To that end, we evaluate the user experience by administering surveys and focus groups, and then we use those responses to generate new strategies and solutions to help the closed-loop system user.”
Testing the “bionic pancreas”
Because the pancreas controls glucose both by releasing insulin to lower glucose levels and by releasing glucagon to raise glucose levels, another approach to closed-loop control is to give both insulin and glucagon. Stanford has participated in an NIH-funded, multicenter study that is testing the “bionic pancreas” developed at Boston University. This system has the potential to eliminate the need for carbohydrate counting before meals while also preventing hypoglycemia through the provision of glucagon. The Lancet recently published an article on this study.
David Maahs, MD, the new division chief of pediatric endocrinology at Packard Children’s, said the program will continue “paving the way for better care, not just for our patients at Packard Children’s, but for people with Type 1 diabetes everywhere.”

