
Stanford Report, January 14th, 2011 - by Susan Young
Travel just one millimeter inside the brain and you'll be stepping into the dark.
Standard light microscopes don't allow researchers to look into the interior of the living brain, where memories are formed and diseases such as dementia and cancer can take their toll.
But Stanford scientists have devised a new method that not only lets them peer deep inside the brain to examine its neurons but also allows them to continue monitoring for months.
Stanford researchers have developed a new technique that allows them to monitor the tiny branches of neurons in a live brain for months at a time. Neuroscientists will now be able to monitor the microscopic changes that occur over the course of progressive brain disease.
The technique promises to improve understanding of both the normal biology and diseased states of this hidden tissue.
Other recent advances in micro-optics had enabled scientists to take a peek at cells of the deep brain, but their observations captured only a momentary snapshot of the microscopic changes that occur over months and years with aging and illness.
The Stanford development appears online Jan. 16 in the journal Nature Medicine. It also will appear in the February 2011 print edition.
Scientists study many diseases of the deep brain using mouse models, mice that have been bred or genetically engineered to have diseases similar to human afflictions.
"Researchers will now be able to study mouse models in these deep areas in a way that wasn't available before," said senior author Mark Schnitzer, associate professor of biology and of applied physics.
Because light microscopy can only penetrate the outermost layer of tissues, any region of the brain deeper than 700 microns or so (about 1/32 of an inch) cannot be reached by traditional microscopy techniques. Recent advances in micro-optics had allowed scientists to briefly peer deeper into living tissues, but it was nearly impossible to return to the same location of the brain and it was very likely that the tissue of interest would become damaged or infected.
With the new method, "Imaging is possible over a very long time without damaging the region of interest," said Juergen Jung, operations manager of the Schnitzer lab. Tiny glass tubes, about half the width of a grain of rice, are carefully placed in the deep brain of an anaesthetized mouse. Once the tubes are in place, the brain is not exposed to the outside environment, thus preventing infection. When researchers want to examine the cells and their interactions at this site, they insert a tiny optical instrument called a microendoscope inside the glass guide tube. The guide tubes have glass windows at the ends through which scientists can examine the interior of the brain.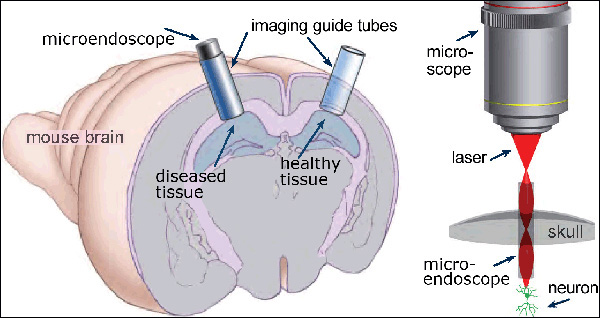
"It's a bit like looking through a porthole in a submarine," said Schnitzer.
The guide tubes allow researchers to return to exactly the same location of the deep brain repeatedly over weeks or months. While techniques like MRI scans could examine the deep brain, "they couldn't look at individual cells on a microscopic scale," said Schnitzer. Now, the delicate branches of neurons can be monitored during prolonged experiments.
To test the use of the technique for investigating brain disease, the researchers looked at a mouse model of glioma, a deadly form of brain cancer. They saw hallmarks of glioma growth in the deep brain that were previously known in tumors described as surficial (on or near the surface).
The severity of glioma tumors depends on their location. "The most aggressive brain tumors arise deep and not superficially," said Lawrence Recht, professor of neurology and neurological sciences. Why the position of glioma tumors affects their growth rate isn't understood, but this method would be a way to explore that question, Recht said.
In addition to continuing their studies of brain disease and the neuroscience of memory, the researchers hope to teach other researchers how to perform the technique.
The first three authors of the paper (all of whom contributed equally to the study) are Robert Barretto, a former doctoral student in biophysics and now a postdoctoral researcher at Columbia University Medical Center; Tony Ko, a former postdoctoral researcher in the Department of Biology; and Jung. Also contributing to the work – and listed as authors – are Tammy Wang, a former undergraduate in biomedical engineering; George Capps and Allison Waters, both former undergraduates in biology; and Yaniv Ziv and Alessio Attardo, both postdoctoral researchers in biology.
The project was supported by funding from the National Institute on Drug Abuse, the National Institute of Neurological Disorders and Stroke, the National Cancer Institute and Mauna Kea Technologies. Robert Barretto was supported by the Stanford-NIH Biophysics Program, and Yaniv Ziv was supported by Machiah and Rothschild postdoctoral fellowships.

