Inside Stanford Medicine - September 21st, 2009 - by Bruce Goldman
Imagine waving a magic wand over a region of a person’s body to determine if he has early signs of cancer. Researchers at the Stanford University School of Medicine have shown, in a preclinical study, that ultrasound energy delivered to specific body regions can trigger the release of biochemical disease markers from tissues, making those markers more detectable as well as helping to pinpoint their source. This relatively simple and noninvasive method may enable earlier detection of diseases, such as cancer, well before obvious symptoms emerge.
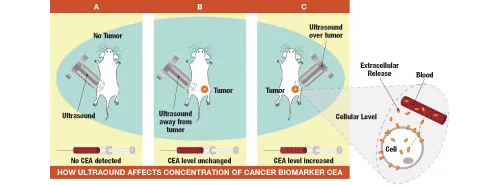
In the study, published online Sept. 21 in the journal Proceedings of the National Academy of Sciences, a team led by Gary Glazer, MD, professor and chair of radiology, found that applying ultrasound energy to a line of tumor cells in culture enhanced those cells’ release into the culture medium of a telltale substance, or biomarker, that distinguishes tumor cells from healthy ones. Using an animal model, they were also able to show that blood concentrations of that biomarker rose significantly when ultrasound energy was directed to tumor sites but, importantly, not when the ultrasound beam was focused on non-tumor-bearing tissues.
Ultrasound energy delivered to specific body regions can trigger the release of biochemical disease markers from tissues, making those markers more detectable as well as helping to pinpoint their source: this relatively simple and noninvasive method may enable earlier detection of diseases, such as cancer, well before obvious symptoms emerge.
“Tumors are often not detected until they become sizable enough to cause recognizable symptoms or to be observed themselves as masses on an X-ray or CT scan,” said Glazer. “By then, the prognosis is much poorer than it might have been had the detection occurred earlier.”
Cancer cells are known to produce substances not normally made by healthy cells and to release quantities of those telltale cancer-specific biomarkers into the blood. Scientists have identified many biomarkers characteristic of particular tumor types or of different cancer stages. In principle, this should allow relatively noninvasive early detection of tumors by monitoring patients’ blood for various cancer biomarkers. But only a handful of them have found their way into clinical practice. One is PSA, an only somewhat accurate early marker of prostate cancer. Another, more precise biomarker, CEA, is used clinically to detect colon cancer.
There are two persistent impediments to the use of biomarkers for early detection of cancer, said Aloma D’Souza, PhD, a research scientist in Glazer’s laboratory and the study’s first author. “Their presence in the patient’s blood is at such low levels — particularly in early-stage disease — that finding them amid all the background noise is very difficult,” she said. “And even when you do find a biomarker, you still don’t know exactly where in the body it came from.”
While circulating levels of some biomarkers may ordinarily be too small for reliable detection, Glazer explained, “the cells that produce them carry multiple copies — up to millions — of these substances on their cell membranes or inside, in their cytoplasm.” At the right frequencies, he said, ultrasound perturbs cells’ outer membranes, permitting larger amounts of biomarkers to be spilled into the bloodstream.
For their study, the investigators first applied ultrasound to cultured colon-cancer cells and showed that doing so caused those cells to release large amounts of CEA into the surrounding culture medium. Then they optimized the ultrasonic energy level to trigger substantial CEA release from the cells without killing large numbers of them. (An important ultimate clinical objective is to be able to find a patient’s tumor without killing adjacent healthy tissue during the search.)
In the next round of experiments, the researchers turned to an animal model: mice with impaired immune systems, which thus can accept implants of foreign tissue, including human tumors. First they measured CEA levels in the blood of the immune-impaired mice for use as a baseline reference. Then, they implanted CEA-producing tumor cells into the mice. Once the cells had engrafted to form tumors, ultrasound beams were directed at both tumor-bearing and, for comparison, tumor-free sites. Ultrasound energy aimed at tumors significantly increased the mice’s blood levels of CEA compared with the levels observed either before tumor cells had been implanted or when the beam was applied to non-cancerous sites in the tumor-bearing animals.
“The application of low-frequency ultrasound to tumor cells caused a significant release of a well-established tumor biomarker into the blood,” said D’Souza. This release occurred only when ultrasound was applied directly to the tumor site.
“We think applications of this approach could come very soon,” she said. “We don’t see significant regulatory impediments to its integration into clinical practice, as ultrasound is widely used already in the clinic. Of course, it will be necessary to optimize the technique for use in humans. And it won’t work for all tumor types — lung or bone-marrow cancers, for example — because ultrasound is impeded by bony structures and air-filled zones in the body. And, while we’ve established proof of principle for a single biomarker, CEA, other biomarkers may prove more difficult to measure this way.”
Still, said Sanjiv Gambhir, MD, PhD, professor of radiology, “this moves us one step closer to solving a key part of the early cancer detection problem, so that we may not have to wait for tumors to grow further in order to detect their presence.” Gambhir, who is also director of Stanford’s Molecular Imaging Program and of the Canary Center for Cancer Early Detection, shares senior co-authorship of the paper with Glazer.
Glazer said the technique could be beneficial for more than the treatment of cancer. “Many common diseases can take decades to develop,” he said. “What if we could detect them a year, or several years, earlier? Some common diseases might not even exist as we know them today if we could detect and treat them that much sooner.”
Other study co-authors were Kim Butts Pauly, PhD, professor of radiology; Samira Guccione, PhD, assistant professor of radiology; and staff scientists Jeffrey Tseng, MD (now at Santa Clara Valley Medical Center in California) and Jarrett Rosenberg, PhD, of the radiology department. Funding for the study was supplied by grants from the Lucas Brothers Foundation, National Institutes of Health, National Cancer Institute, Canary Foundation and Stanford’s Department of Radiology.

