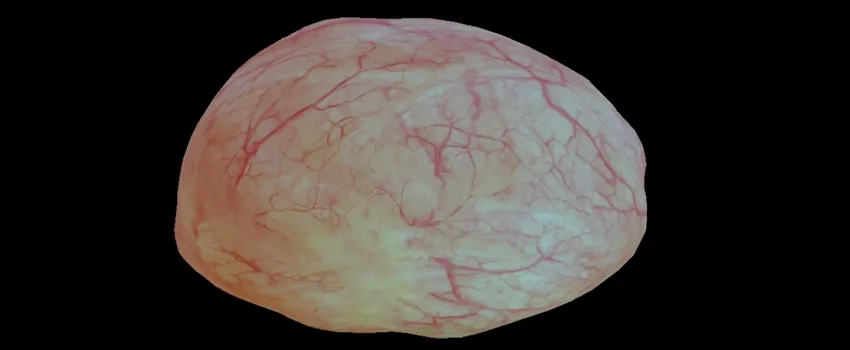
Screenshot from video by Kristen Lurie. Audrey Bowden, assistant professor in the Department of Electrical Engineering, is a co-senior author of a paper about modeling the bladder with advanced computer imaging technology.
Stanford News - March 16th, 2017 - by Jackie Flynn
The way doctors examine the bladder for tumors or stones is like exploring the contours of a cave with a flashlight. Using cameras attached to long, flexible instruments called endoscopes, they find that it’s sometimes difficult to orient the location of masses within the bladder’s blood vessel-lined walls.
This could change with a new computer vision technique developed by Stanford researchers that creates three-dimensional bladder reconstructions out of the endoscope’s otherwise fleeting images. With this fusion of medicine and engineering, doctors could develop organ maps, better prepare for operations and detect early cancer recurrences. See video here.
“The beauty of this project is that we can take data that doctors are already collecting,” said Audrey Bowden, assistant professor in the Department of Electrical Engineering. She is a co-senior author on a recent paper published in Biomedical Optics Express that describes the method.
Bladder cancer has among the highest recurrence rates of any cancer. From 50 percent to 70 percent of tumors return after removal, according to Joseph Liao, an associate professor of urology at the School of Medicine and co-senior author on the paper. Being able to see each patient’s bladder as a three-dimensional model could improve surgical planning and monitor cancer recurrence.
“Endoscopy of the bladder, called cystoscopy, is an integral part of cancer management. Anything you can do to improve endoscopy is helpful,” Liao said. “Surgeons are always looking for better ways to see cancer in order to remove it more effectively.”
Broadly applicable technique
One of the technique’s advantages is that doctors don’t have to buy new hardware or modify their techniques significantly. Through the use of advanced computer vision algorithms, the team reconstructed the shape and internal appearance of a bladder using the video footage from a routine cystoscopy, which would ordinarily have been discarded or not recorded in the first place.
“In endoscopy, we generate a lot of data, but currently they’re just tossed away,” said Liao.
Although the team developed the technique for the bladder, it could be applied to other hollow organs where doctors routinely perform endoscopy, including the stomach or colon. “We were the first group to achieve complete 3D bladder models using standard clinical equipment, which makes this research ripe for rapid translation to clinical practice,” said Kristen Lurie, lead author on the paper, a recent PhD graduate and postdoctoral fellow at Stanford, and a current software engineer at Google.
According to Liao, these three-dimensional images could help doctors prepare for surgery. Lesions, tumors and scars in the bladder are hard to find, both initially and during surgery.
“Sometimes you don’t have a sense – where was I in the bladder?” Liao said. Seeing a three-dimensional rendering of an organ before operating, like having a map before embarking on a trip, could make the procedure easier for doctors. Other potential applications include using the 3D reconstruction as a visual medical record.
Accurate rendering
To test the accuracy of their bladder reconstruction, the team first created a model based on endoscopy images taken in a 3D-printed bladder, known as a tissue phantom. Because the details of the tissue phantom are known, the researchers could directly compare their rendering to the real thing. According to Bowden, tissue phantoms provide a standard for biological modeling analysis. The team found that its three-dimensional rendering matched the tissue phantom with few errors.
This technique is the first of its kind and still has room for improvement, the researchers said. Primarily, the three-dimensional models tend to flatten out bumps on the bladder wall, including tumors. With the model alone, this may make tumors harder to spot. The team is now working to advance the realism, in shape and detail, of the models.
Future directions, according to the researchers, include using the algorithm for disease and cancer monitoring within the bladder over time to detect subtle changes, as well as combining it with other imaging technologies.
“The technology has immense potential, not to mention the fun factor to be able to navigate around a virtual organ,” Liao said.
This research was supported by an NSFGRFP fellowship and the Max Planck Center for Visual Computing and Communication.
Bowden is also a member of Stanford Bio-X and of the Stanford Child Health Research Institute. Liao is also a member of Stanford Bio-X and of the Stanford Cancer Institute. Other authors include Roland Angst and Dimitar V. Zlatev.


